Who do we Distribute to?
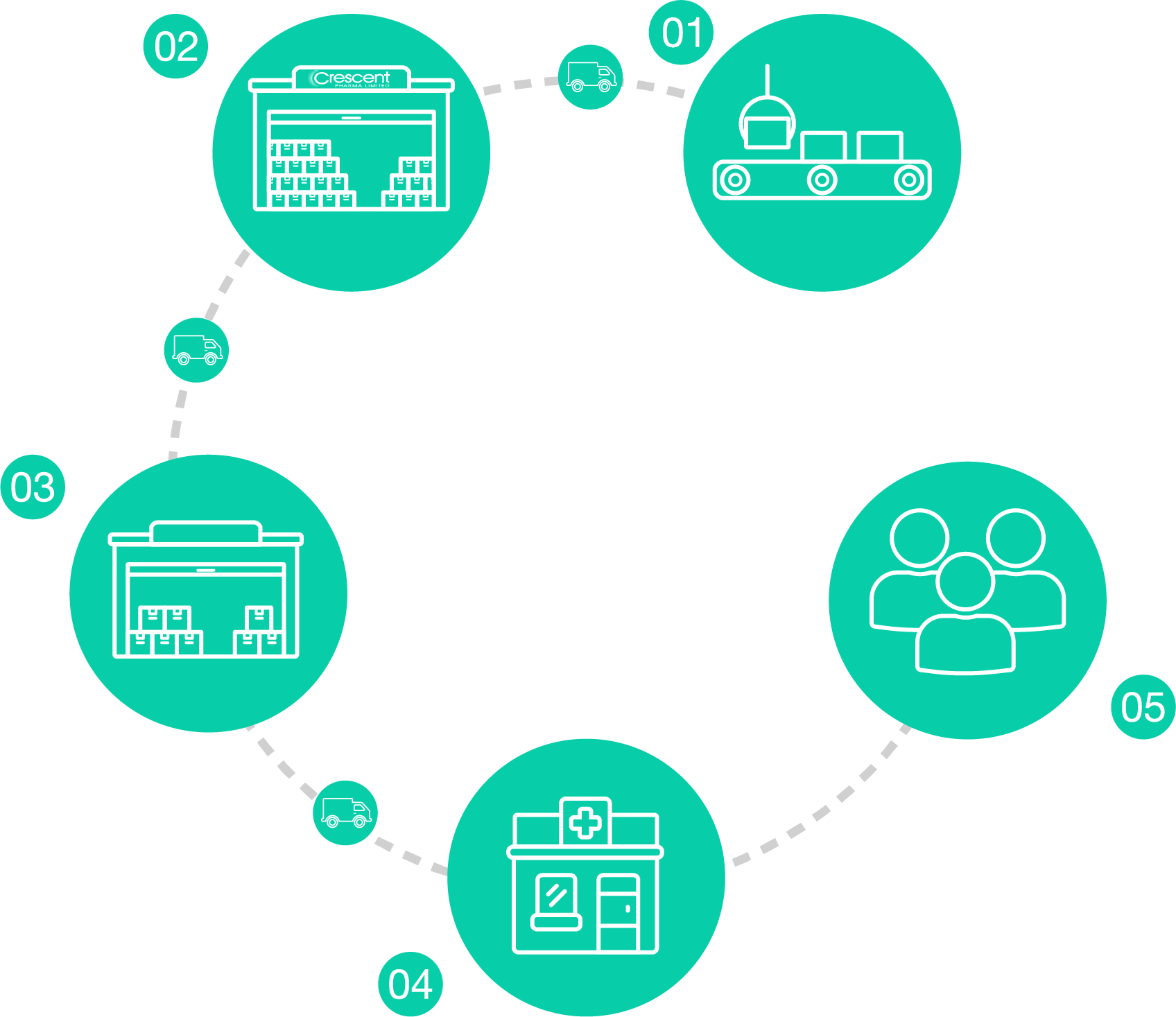
01
Manufacture
Crescent source and produce licensed medicines from regulatory approved sites.
02
Storage
Crescent Pharma store all of their products in MHRA approved warehouses and distribution centres
03
Wholesalers & Pharmacy groups
Crescent Pharma distributes its products direct from our depots using temperature-controlled transport to pharmaceutical wholesalers and pharmacy groups.
04
Pharmacy
Pharmacies order our products directly from Pharmaceutical wholesalers
05
Patients
The Pharmacies then dispense our products to patients
Crescent Pharma only sell to licensed suppliers. Crescent Pharma do not sell products directly to the general public.
packs distributed in 2022 to customers all over the UK
0
million
pallets in our approved temperature-controlled facilities here in the UK
0
Crescent Pharma packs have been delivered to patients since 2019
0
million
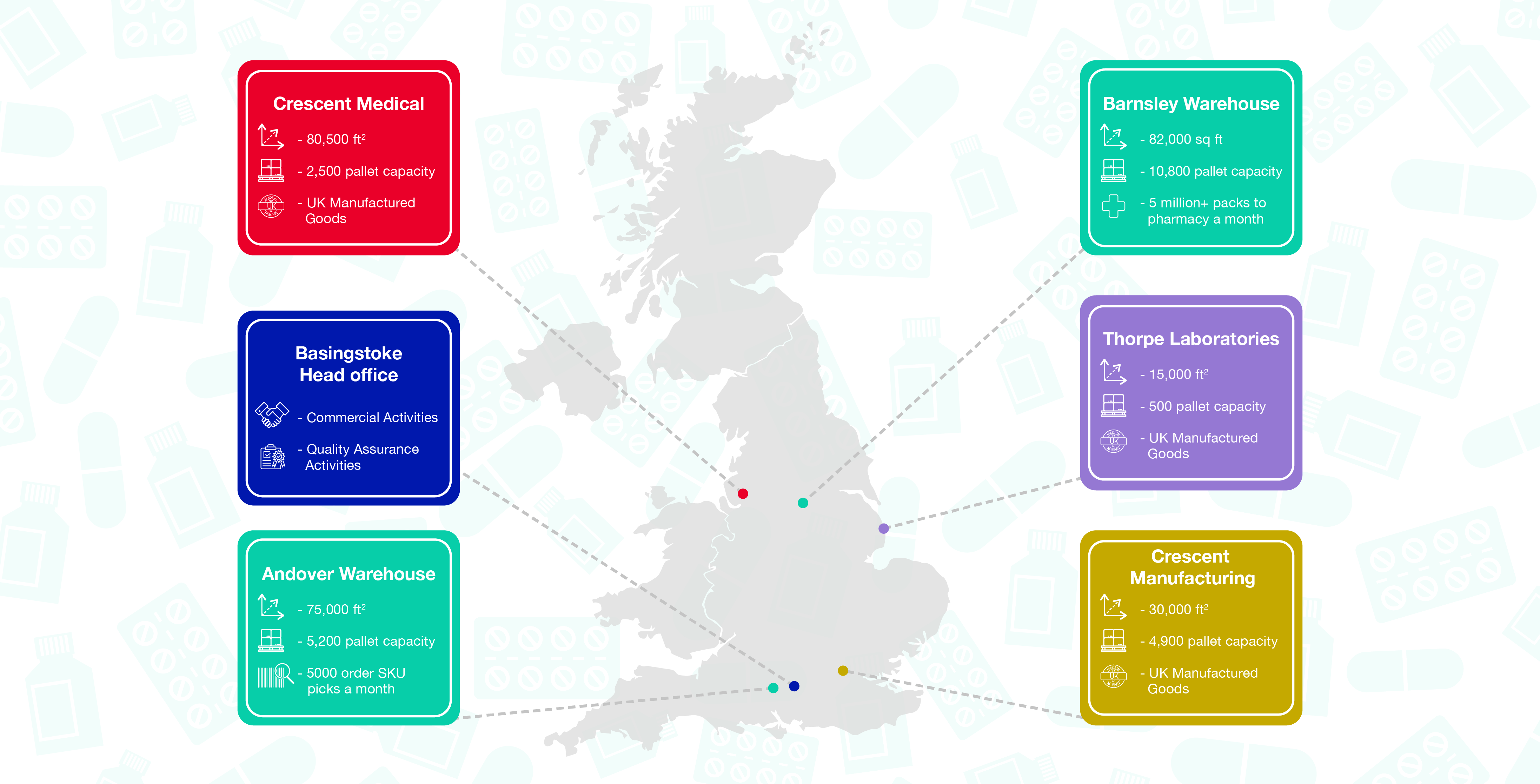
Our distribution Network
Crescent Pharma own three dedicated distribution centres as well as working with key MHRA approved storage sites to service the UK patient demand, housing over 45,000 pallets of medicine. Crescent Pharma’s internal network houses over 22K pallets, covering the length and depth of the UK.
Crescent Pharma also work with select partners to hold an additional 25k pallets of stock at external sites based in the UK.









The UK Pharmaceutical Distribution Network
In the UK, pharmaceutical distribution involves the process of getting Crescent Pharma’s products to various points of sale, including wholesalers, distributors, pharmacies, and other healthcare providers. This distribution network is complex and plays a crucial role in ensuring the availability and accessibility of pharmaceutical products to patients and healthcare professionals.
To maintain the integrity and safety of the distribution process, the pharmaceutical industry in the UK is highly regulated. The Medicines and Healthcare products Regulatory Agency (MHRA) oversees the licensing, safety, and quality control of pharmaceutical products. They enforce regulations and standards to ensure that products are manufactured, stored, transported, and dispensed in a manner that meets strict quality and safety guidelines.

01
Wholesalers
Wholesalers serve as intermediaries between Crescent Pharma and other entities in the distribution chain. They purchase products in bulk from the manufacturer, including Crescent Pharma, and store them in warehouses. Wholesalers then distribute these products to various customers, such as pharmacies, hospitals, and other healthcare providers. They play a vital role in ensuring a steady supply of pharmaceutical products to meet the demands of the market.
02
Distributors
Distributors also play a significant role in the pharmaceutical distribution process. They may specialize in specific regions or types of products and act as intermediaries between wholesalers and pharmacies or healthcare providers. Distributors help streamline the supply chain by efficiently delivering products to their designated destinations.
03
Pharmacies
Pharmacies are essential points of sale in the pharmaceutical distribution network. They dispense medications directly to patients and are often the final stop before products reach the end-users. Pharmacies procure pharmaceutical products from wholesalers, distributors, or directly from manufacturers like Crescent Pharma. They play a crucial role in providing patient information, prescription management, and ensuring safe and accurate medication dispensing.
04
Healthcare Providers
In addition to wholesalers, distributors, and pharmacies, other healthcare providers may also be part of the distribution network. This can include hospitals, clinics, nursing homes, and other healthcare facilities. These entities procure pharmaceutical products for their own use or to dispense to patients within their care.
Pharmaceutical Distribution and Regulation in the UK
Pharmaceutical distribution in the UK follows the principles of Good Distribution Practice (GDP) guidelines. GDP is a set of quality assurance standards and requirements that ensure the proper storage, transportation, and distribution of pharmaceutical products. These guidelines are designed to maintain the integrity and quality of medicines throughout the distribution chain, ultimately safeguarding patient safety.
By following GDP guidelines, the pharmaceutical distribution network in the UK aims to guarantee the quality, safety, and efficacy of medicines throughout the supply chain. These guidelines help prevent counterfeit or substandard products from entering the market and ensure that patients receive reliable and effective medications.
The implementation of GDP guidelines begins with the manufacturer
Crescent Pharma must adhere to strict quality control measures during the production, packaging and transportation of pharmaceutical products. They must maintain appropriate facilities, equipment, and processes to ensure the products' safety, efficacy and quality.
License holders, Wholesalers, Pharmacies and distributors
Play a crucial role in GDP-compliant pharmaceutical distribution. They are responsible for receiving, storing, and distributing pharmaceutical products to pharmacies, hospitals, and other healthcare providers. These entities must have proper storage conditions, such as temperature-controlled facilities, to maintain the products' stability and prevent any degradation or contamination.
Robust quality management systems
GDP guidelines require license holders, wholesalers and distributors to establish robust quality management systems. This includes documenting and maintaining comprehensive records of product movements, batch numbers, expiry dates, and storage conditions. The guidelines also emphasise the need for proper documentation of transportation processes, including temperature monitoring during transit.
Regulatory Authorities
In the UK, wholesalers and distributors must obtain the necessary licenses and authorisations from the regulatory authorities, such as the Medicines and Healthcare products Regulatory Agency (MHRA). These licenses ensure that the entities meet the GDP standards and comply with regulatory requirements. To ensure compliance with GDP guidelines, regulatory authorities conduct inspections and audits of wholesalers, distributors, and pharmacies. They assess the facilities, procedures, documentation, and quality management systems to verify adherence to the required standards. Non-compliance can result in penalties, sanctions, or the suspension of licenses.
Pharmacies and Healthcare providers
Pharmacies and other healthcare providers receiving pharmaceutical products must also adhere to GDP guidelines. They should have appropriate storage conditions, ensure proper handling and transportation, and maintain accurate records of product receipt, storage, and dispensing. They are responsible for checking the integrity and authenticity of the products, verifying the expiry dates, and managing any product recalls or returns as per the guidelines.
